If you’re a clinician who needs to translate evidence into practice or are a researcher this is essential reading.
Systematic reviews serve a number of cruical roles when it come to understanding the state of knowledge on a topic and identifying future research priorities. Often they are the only way we can address questions that couldn’t be answered by individual studies alone and they are a vital resource for clinicians, researchers, patients and policy makers alike. However, they only achieve this role when they are transparent, accurate and complete including the why’s and what’s of the review itself.
To ensure a systematic review is valuable to users, authors should prepare a transparent, complete, and accurate account of why the review was done, what they did, and what they found…
The Preferred Reporting Items for Systematic review and Meta-Analyses (PRIMSA) statement is a guideline designed to ensure that systematic reviews are accurate, complete and transparent. It’s aim is to combat the poor reporting of systematic reviews and comprises of a checklist of 27 items recommended for reporting in systematic reviews. Since it’s creation in 2009 it has been widely endorsed and cited over 60,000 times. To say it is essential is an uderstatement the statement is practically synonymous with systematic review.
Improve Your Confidence Interpreting Research
Since 2009 many research and techology innovations have taken place including new research methods to assess risk of bias and machine learning to identify patterns which make areas of PRISMA less useful and relevant. The language used within systematic review research has also changed with a shift from assessing quality to assessing certainty. To make sure these, as well as numerous other advances, the PRISMA 2009 statement needed to be updated.
Development of PRISMA 2020
There is a full description of how the updated statement was developed which was published in the Journal of Clinical Epidemiology in early Feb 2021. The journey began in 2017 where a group of international experts in systematic review methodology was established to lead the update.
A 4-step process was used to update the guidance. First over 60 documents were reviewed to generate modifications to the 2009 statement. 110 systematic review methodologists and editors then completed a 3-part survey about the suggested modifications. The results of both steps 1 and 2 were discussed at a 21-member in-person meeting held in Edinburgh, Scotland, in 2018. After the meeting the 4th and final step took place which involved drafting and iteratively changes to the statement, checklist, flow diagram and supplementary information until the final document was completed in Spring 2020.
How To Use PRISMA 2020 & Understanding Scope
PRISMA 2020 doesn’t tell researchers how to conduct a systematic review rather it aims to aid in planning the research to ensure all the recommended information is captured. Likewise PRISMA doesn’t assess the methodological quality of a review, but rather the appropriateness of the methods adopted and the trustworthiness of findings. It’s a hallmark of a robust and transparent review process which facilitates replication of results and ultimately improve the certainty of research findings.
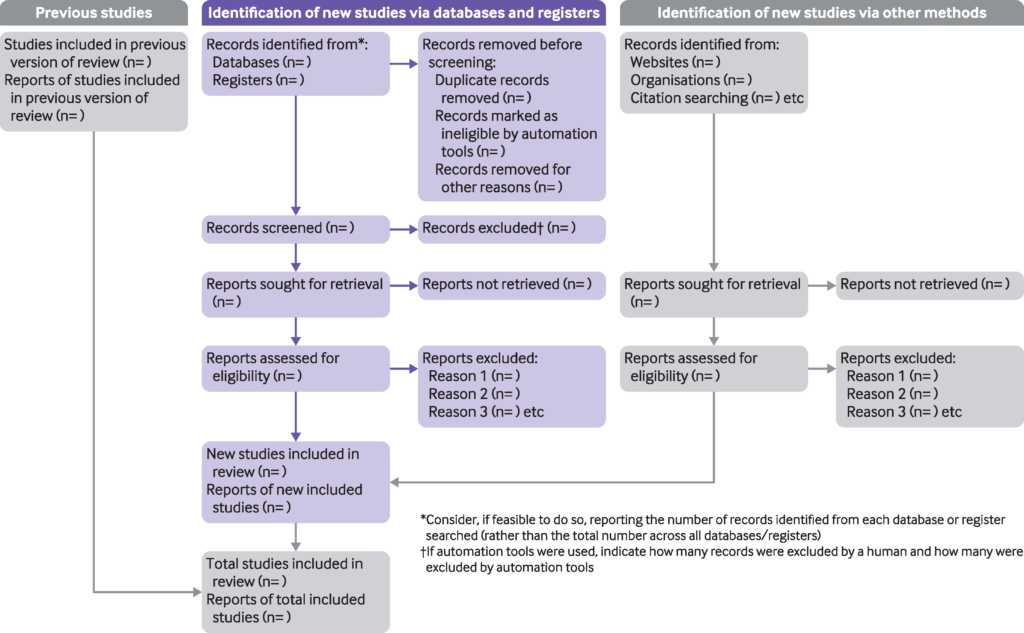
The checklists, explanation and flow diagram have all been updated and from now on the 2009 versions should not be used. This includes all PRISMA extensions including abstracts and protocols which now align with the statement as previously they were updated in 2015.
Quick Summary of Updated PRISMA 2020 Guidance
There has been criticism recently of not having enough research methodology expoloration in undergraduate education. Incorporating the new PRISMA 2020 statement into undergraduate physiotherapy curricula would be a strong addition to understanding the importance of transparent and replicable research methodologies. It is straightforward in it’s concept and the supplementary checklists are user friendly making it applicable to anyone of any research experience level.
Physiospot features easy-to-digest summaries of many systematic reviews and this is because of their transferability and robust methodology meaning lessons can be translated to clinical practice with confidence. Without PRISMA this wouldn’t be such an easy process and the updated statement only makes the role of systematic reviews more important.